The Common Vein Copyright 2007
Ashley Davidoff MD
Jessica Humphries
Ganesh Athappan MD
Embryology
Ashley Davidoff

Ashley Davidoff

Ashley Davidoff
Ashley Davidoff
Ashley Davidoff
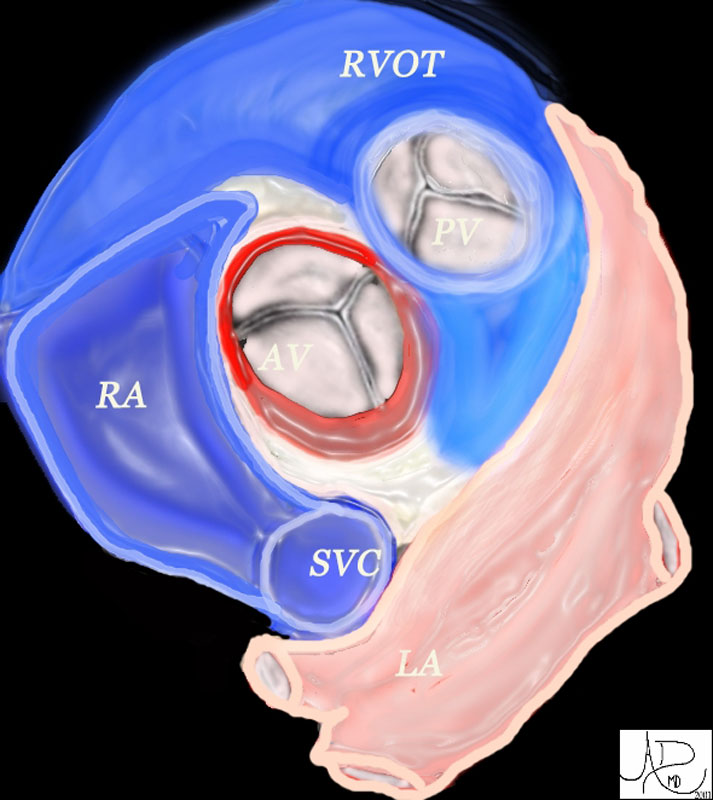
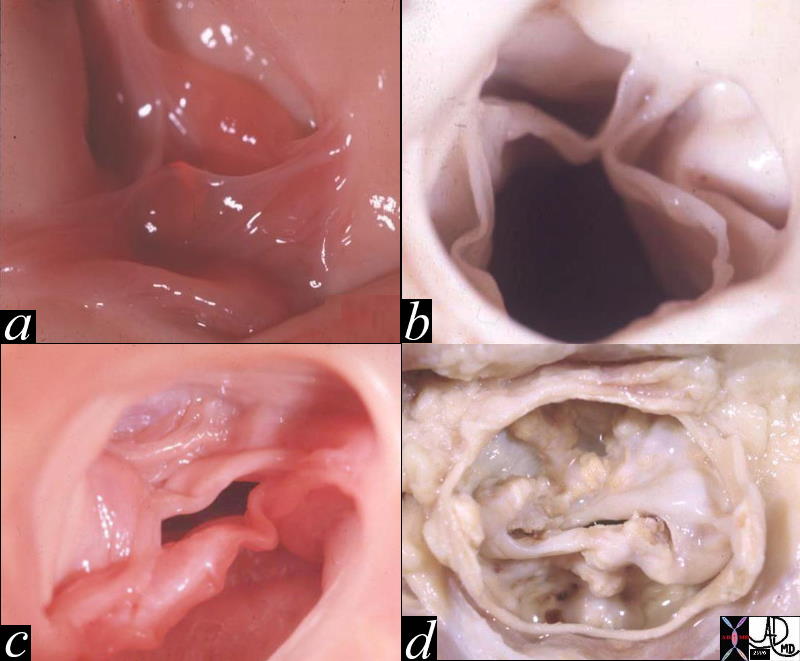
The aortic valve (AV) lies central to many structures including the right ventricular outflow tract (RVOT), pulmonary valve (PV), left atrium (LA), right atrium (RA), and superior vena cava (SVC). aorta from above. In this diagram, the commissures are overlaid in green, the crescent shaped lunulae reflect the free edge of the leaflets and are overlaid in yellow, and the nodules of Arantii are overlaid in orange. The sinuses of Valsalva are like cups and are positioned between the free edges. (gray)
Davidoff art Ashley Davidoff MD TheCommonVein.net 47681

The aortic valve (AV) lies central to many structures including the right ventricular outflow tract (RVOT), pulmonary valve (PV), left atrium (LA), right atrium (RA), and superior vena cava (SVC). aorta from above. In this diagram, the commissures are overlaid in green, the crescent shaped lunulae reflect the free edge of the leaflets and are overlaid in yellow, and the nodules of Arantii are overlaid in orange. The sinuses of Valsalva are like cups and are positioned between the free edges. (gray)
47681d07
Ashley Davidoff MD
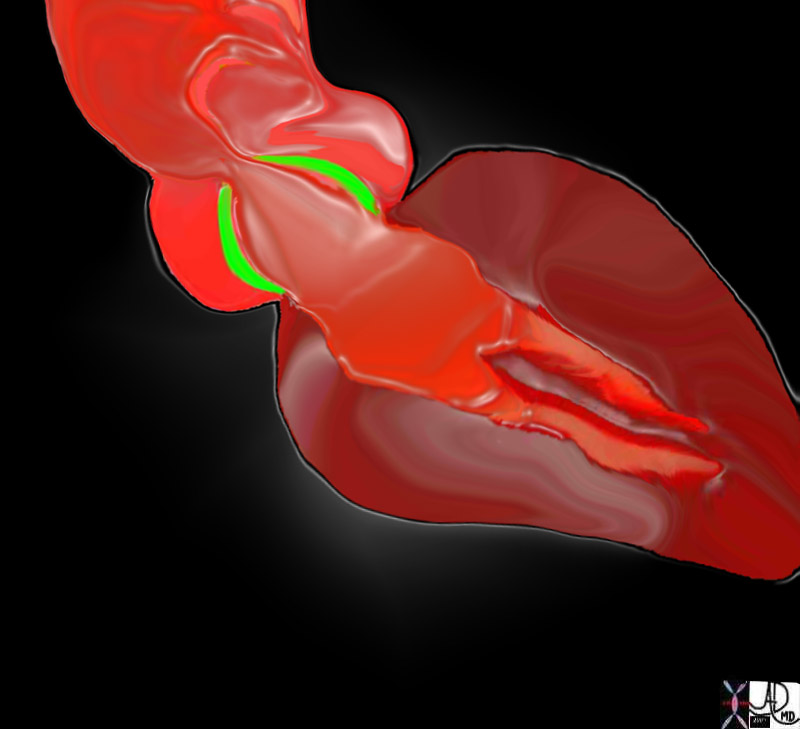
A commissure is the space or area between each anchored leaflet to the aortic wall. They act as support to the base structure of the cusps.

Ashley Davidoff
Keywords:
normal heart cardiac LV MV anterior leaflet fibrous continuity anatomy
01817.800
Keywords:
normal heart cardiac LV MV anterior leaflet fibrous continuity anatomy
15070

32119b02p

Radiographics Bennett
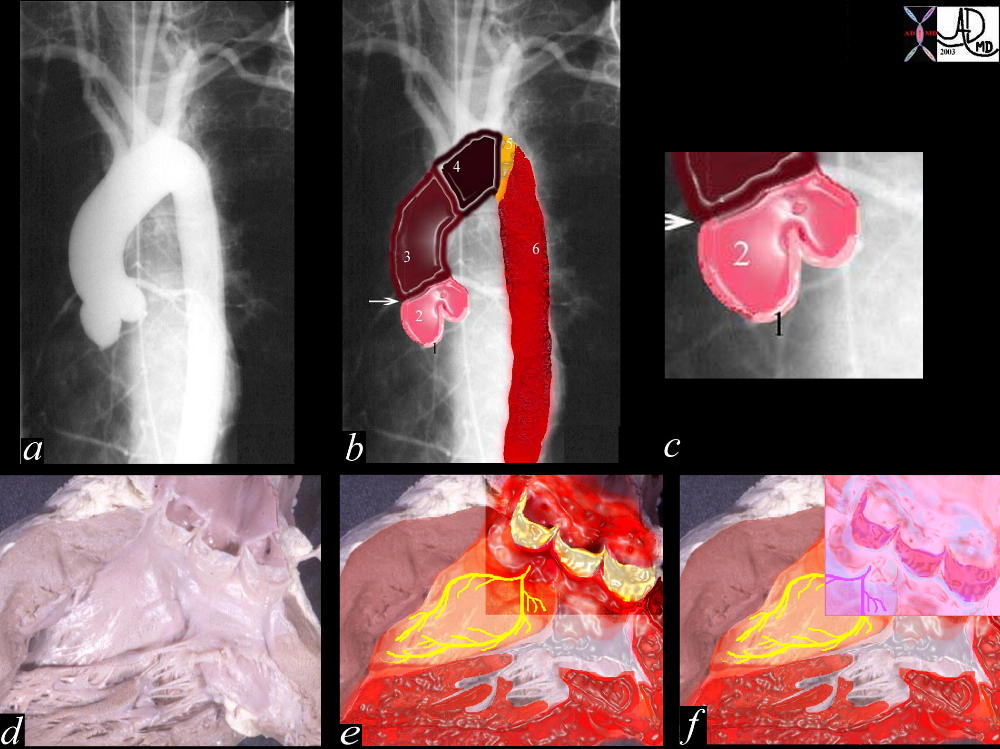
Left Bundle branch in the region of the membranous septum between the non and right coronary cusps
Fibrous continuity of the anterior leaflet of the mitral valve with the left and non coronary cusp Ashley Davidoff

Left Bundle branch in the region of the membranous septum between the non and right coronary cusps
Fibrous continuity of the anterior leaflet of the mitral valve with the left and non coronary cusp Ashley Davidoff 07970e02
Radiographics Bennett

Ashley Davidoff
Ashley Davidoff
Ashley Davidoff
Definition
The aortic valve is part of the circulatory system and is one of the four valves of the heart.
It is located between the left ventricle and the base of the aorta and is structurally characterized by three leaflets in a semi lunar shape.
The function of the aortic valve is to open passively during systole and close during diastole.
Common diseases of the aortic valve include calcified aortic stenosis, aortic regurgitation, and bacterial endocarditis, which sometimes progresses to ring abscess. Bicuspid aortic valve is one of the most common congenital cardiac abnormalities.
Commonly used diagnostic procedures for these diseases include echocardiography and CT and MRI scans.
Diseases of the aortic valve are usually treated with surgery, particularly aortic valve replacement.
Aortic Valve
The aortic valve is one of the four valves of the heart, located between the left ventricle and the base of the aorta. It is characterized structurally by three leaflets in a semi lunar shape. The root of the valve is attached to the aorta in a curvilinear manner. The one way valve opens passively during systole and closes during diastole. Common diseases include calcific aortic stenosis, aortic regurgitation and bacterial endocarditis which sometimes progresses to ring abscess. Bicuspid aortic valve is one of the most common congenital cardiac abnormalities. Most entities of the aortic valve are best and easily diagnosed by echocardiography. CTscan and MRI are useful as well. Surgical treatment, particularly aortic valve replacement is common.
Structure
The aortic valve is attached to the annulus. The valve consists of three semilunar cusps, larger, thicker, and stronger than those of the pulmonary valve. Each cusp has a thickened nodule (aka: nodulus, corpus Arantii, nodulus valvulae semilunaris, Arantius’ nodule, Bianchi’s nodule, Morgagni’s nodule) in the center of its free margin. These nodules are thicker and more prominent than those of the pulmonary trunk. The points where the attachments of two adjacent cusps are joined are called commissures. There are also thickened bands extending from nodules to commissures. These bands arch in crescentic manner and hence are called lunulae (little moons). They are more distinct than the lunulae of the pulmonary trunk. In addition, there are dilated pockets between the cusps and the aortic wall; each pocket is called an aortic sinuses (aka: sinus of valsalva, sinus aortae, Petit’s sinus) and from the sinuses the coronary arteries originate. In concert, they are approximately the size of a quarter.

The aortic valve (AV) lies central to many structures including the right ventricular outflow tract (RVOT), pulmonary valve (PV), left atrium (LA), right atrium (RA), and superior vena cava (SVC). aorta from above. In this diagram, the commissures are overlaid in green, the crescent shaped lunulae reflect the free edge of the leaflets and are overlaid in yellow, and the nodules of Arantii are overlaid in orange. The sinuses of Valsalva are like cups and are positioned between the free edges. (gray)
Davidoff art Ashley Davidoff MD TheCommonVein.net 47681d07
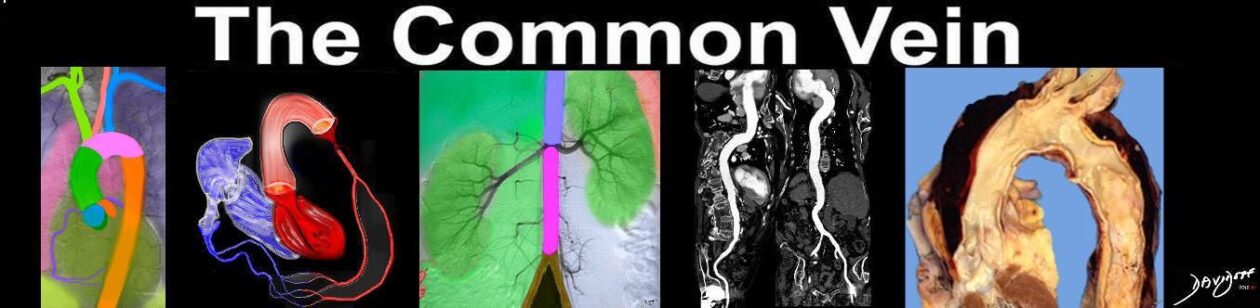
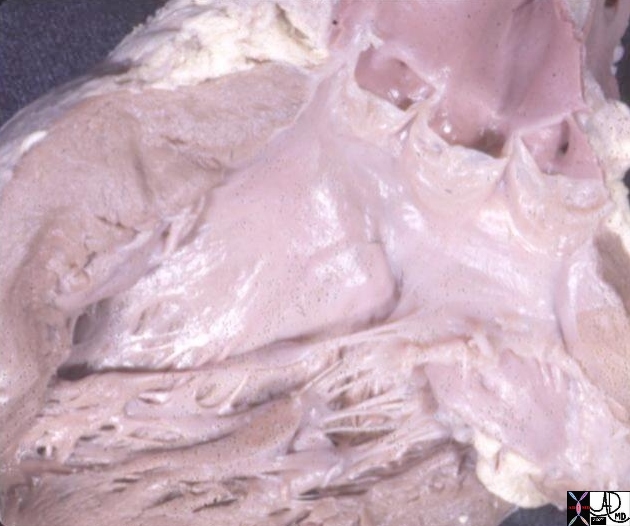
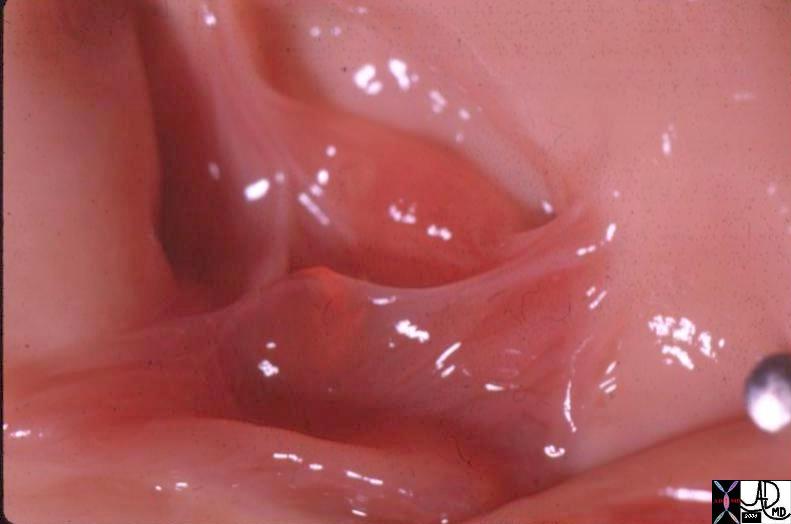 Normal Aortic Valve in the Neonate Normal Aortic Valve in the Neonate |
| 07477 cardiacheart valve aortic valve leaflets normal gross anatomy grosspathology Davidoff MD |
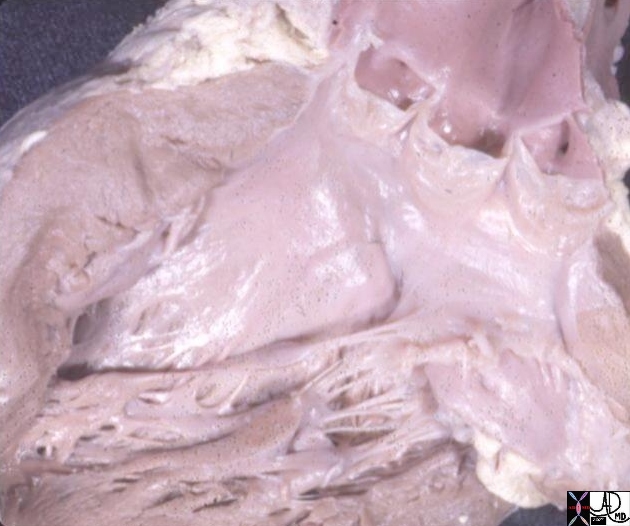 Aortic Valve in the A0dult Aortic Valve in the A0dult |
| The left ventricular outflow tract has been opened showing the three valve leaflets and their associated sinuses. The right and left aortic cusps each contain a coronary ostium The non coronary cusp lies between the left and right cusps, and is without an ostium.
Courtesy Ashley Davidoff MD 07970 |
The aortic valve’s semilunar shape provides a complementary three dimensional structure that can open and close without blood leakage. During systole the valves are forced open by the ejection pressure created by the heart. During diastole, as the pressure starts to fall, the blood fills back into the pockets of the sinuses of Valsalva causing them to distend enabling the leaflets to coapt perfectly so that there is no regurgitation back into the left ventricle.
 Systole (left) and Diastole (right) Systole (left) and Diastole (right)
The Semilunar Cusps are Forced Open in Systole and Close Passively Due to Back Flow of Blood into the Sinuses of Valsalva |
| 32119d04 Courtesy Ashley Davidoff MD |
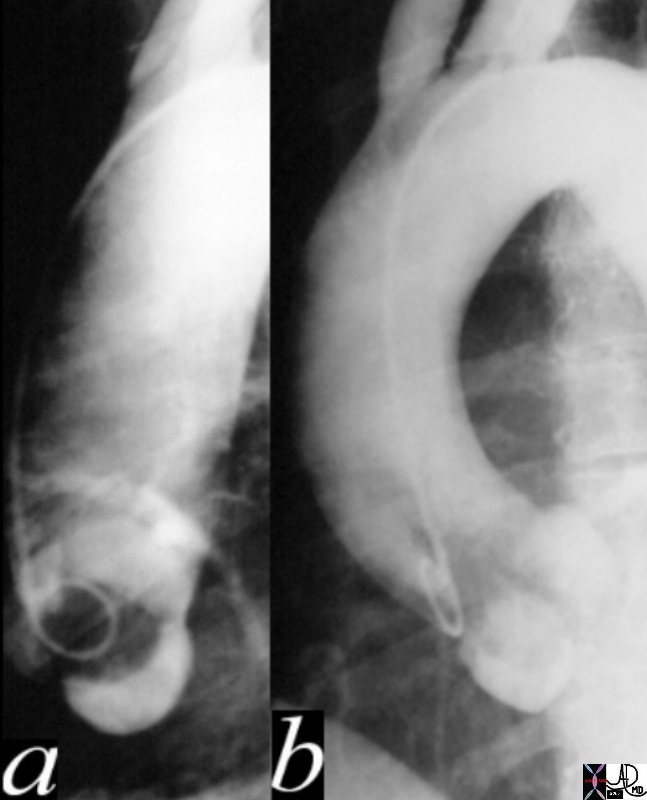
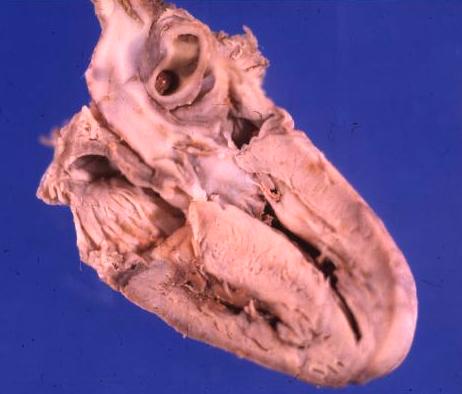
Aortic Valve Open in Systole (a) and Closed in Diasttole (b) |
| 17512c02.800 thorax aortic valve normal anatomy physiology systole diastole aortic valve open closed 3 cusps thoracic aorta ascending aorta Courtesy Ashley Davidoff MD |
Histology and Ultrastructure
The valve is made connective tissue consisting mostly of of collagen, and elastin. There are three layers; the surface layer is called a fibrosa, the spongiosa, and the ventricularis. The fibrosa lies with its surface facing the aortic lumen, while he ventricularis faces the ventricular lumen. The fibrosa is made mostly of collagen that is arranged circumferentially. The leaflets are composed of endocardium and connective tissue reinforced by non-muscular fibers. The aortic valve is similar in structure and function to the pulmonary valve but since it is subjected to the higher pressures of the left ventricle, it is thicker and more fibrous
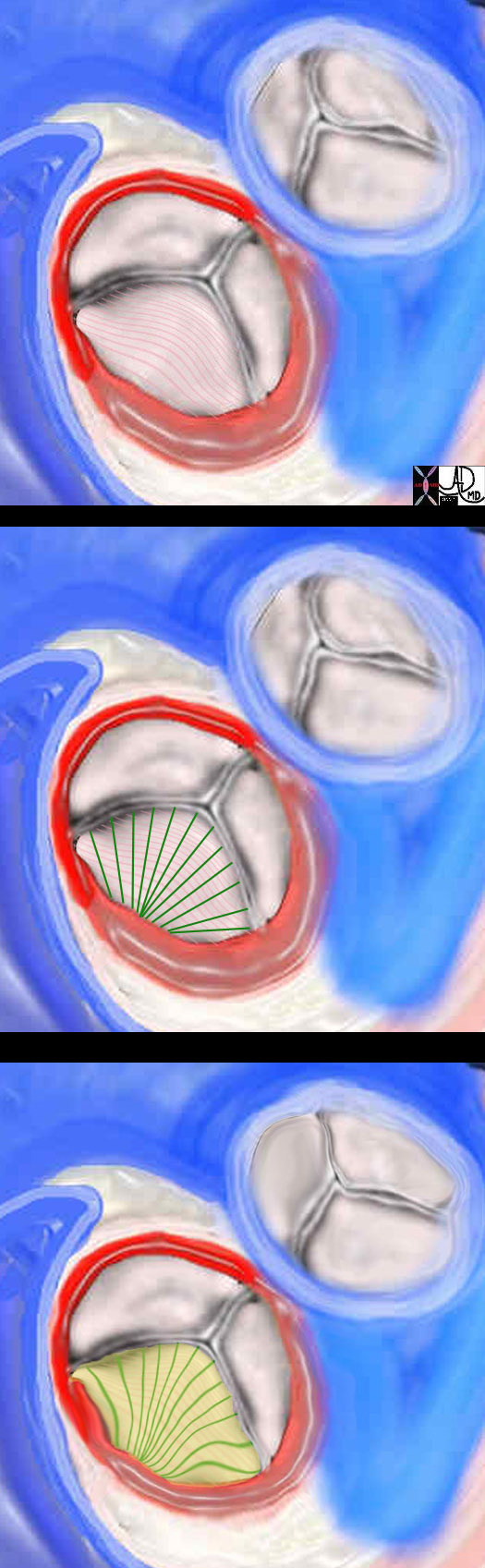
Superficial Fibrosa, Middle Spongiosa, and Deep Ventricularis
There are 3 layers of collagen within the valve. The top image shows the fibrosa layer on the aortic side of the valve. (pink) This layer consists of radially oriented fibres extending from commissure to commissure. It has corrugated form enabling it to stretch during systole. The middle layer is called the spongiosa green and its fibers extend at right angles to the fibers of the fibrosa. On the ventricular side in the lowest image a sheet of tissue called the ventricularis is positioned.
Courtesy Ashley Davidoff Davidoff art TheCommonVein.net 47682d08

Corrugated Superficial Fibrosa, (pink) Middle Spongiosa, (green)
and Deep Ventricularis (yellow)
Courtesy Ashley Davidoff theCommonVein.net Davidoff art 47682eb06.800
The design of the aortic and pulmonary valves is much simpler than the design and structure of the atrioventricular valves (mitral and tricuspid). For example the A-V valves have a varied number, size, and shape, of the component leaflets. In addition they are attached to chordae tendinae and papillary muscles which tether them to the ventricular walls. The aortic and pulmonary valves are not anchored in such a manner, and operate in passive manner, opening and closing according to pressure differences between the ventricles and the great arteries.
Naming the Leaflets
The three leaflets are named according to their position and their relationship to the coronary ostia. Hence there is a left coronary cusp, right coronary cusp and a non coronary cusp. The right cusp lies anterior and to the right, the left lies leftward and slightly posterior, while the non coronary cusp is most posteriorly located and also the most inferiorly.
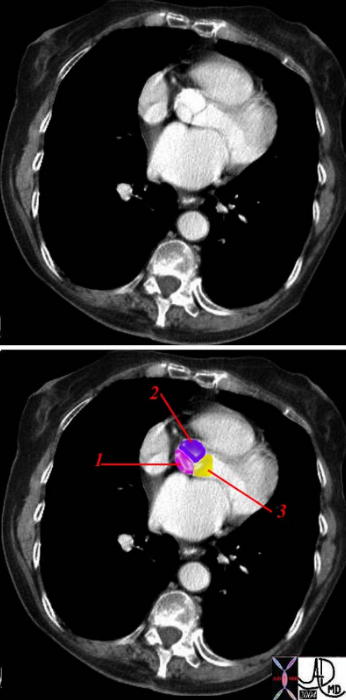
Aortic Cusps |
| The leaflets are named according to their position and relationship to the coronary ostia.
1= non coronary cusp 2= right coronary cusp 3 = left coronary cusp Courtesy Ashley Davidoff MD 38357c02 |
Parts
Structurally the aortic valve complex or aortic root includes the the aortic annulus ,semilunar valve made up of three half moon shaped cusps or leaflets , coronary sinuses and the sinotubular ridge or supra aortic ridge.
The aortic annulus, scalloped in shape , is a fibrous structure that attaches the aortic root to the left ventricle. It is attached directly to the myocardium in approximately 45% of its circumference and to fibrous structures in the remaining 55% (anterior leaflet of mitral valve and membranous septum)2.
Size
The normal area of the aortic valve is 3-4cms2. When the valve is 1cms2 or less critical stenosis is present and is an indication for surgical replacement. Mild stenosis is present when the gradient is 40mmHg, moderate when less than 60mmHg and severe when the pressure gradient is more than 80mmHg. The valve diameter at slightly more than 3cms is just slightly larger than th main pulmonary artery. The semilunar valves ( aortic and pulmonary) are smaller than the AV valves1
Each leaflet has been described as having a semi lunar shape – ie half a moon. It can also be perceived as having almost a U shape, with both the upper border (free edge) and the lower border (attached to the aortic wall) describing a “U”. Orifice of the aortic valve may be either triangular or circular1.

Davidoff Art Courtesy Ashley Davidoff MD TheCommonVein.net 47682f02.800
Position
Surface anatomy : The aortic valve lies behind and to the left of the sternum opposite the 3rd intercostal space.
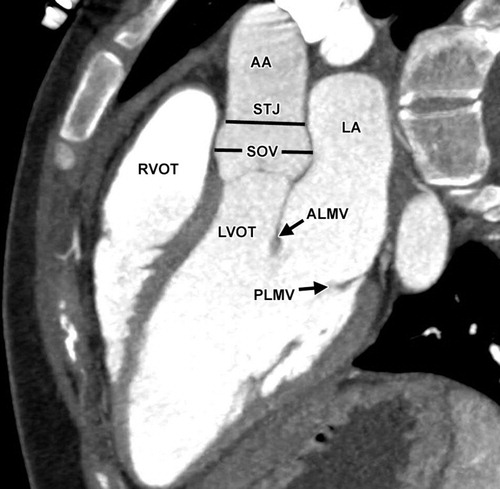
The position of the aortic valve in relation to the mitral valve, its angular relationship to the LVOT, and its position in relation to the pulmonary artery and pulmonary valve are all critical in terms of the embryology, physiology, and pathology. The valve lies between the two low pressure atria, probably one of the most comfortable spots in the body as blood flows in waves, almost simulating the sound of the waves of the ocean. It lies posterior tand to the right of the right ventricular outflow tract and main pulonary artery, and lies below and to the right of the pulmonary valve. It is oriented toward the right shoulder, and tilted so that the left coronary ostium is slightly higher than the right.
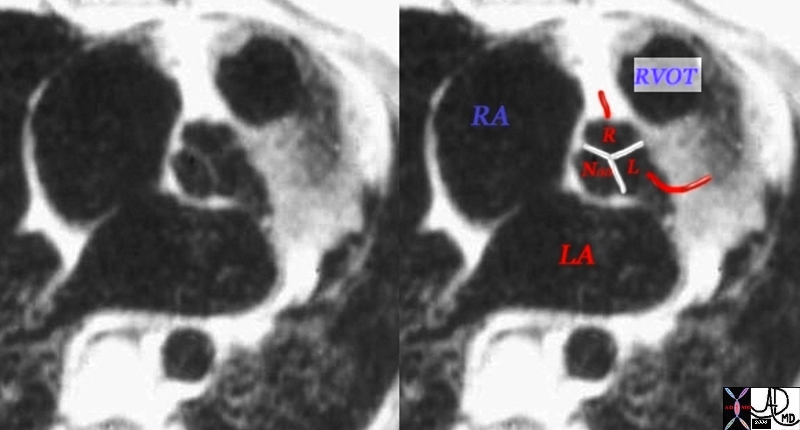
Normal Position and Relationship of the Aorta and Pulmonary Artery |
| 07954bWduo heart cardiac aorta aortic valve infundibulum RVOT right ventricular outflow tract position relation MRI scan Davidoff MD |
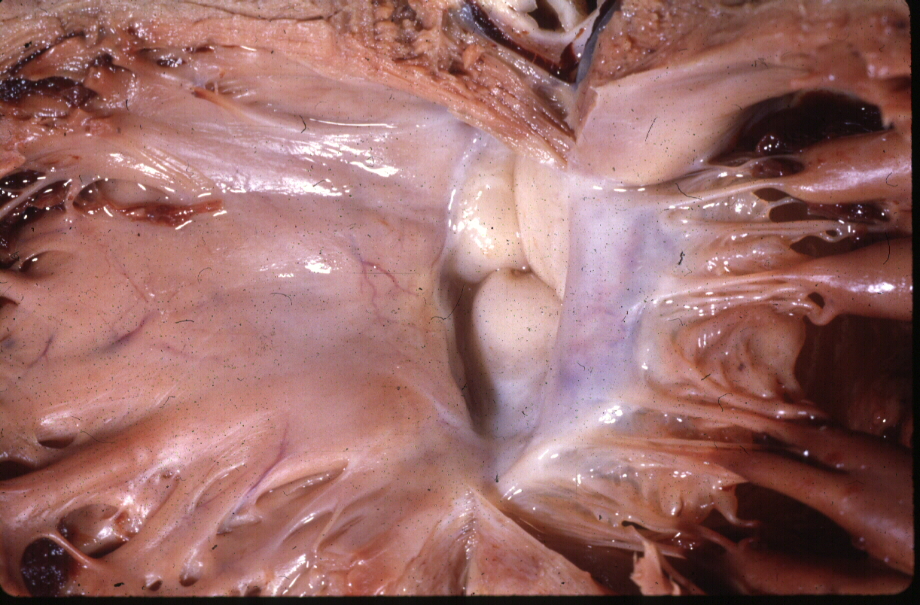
The Relationship of the Aortic Valve to the Mitral Valve and Other Parts of the Heart
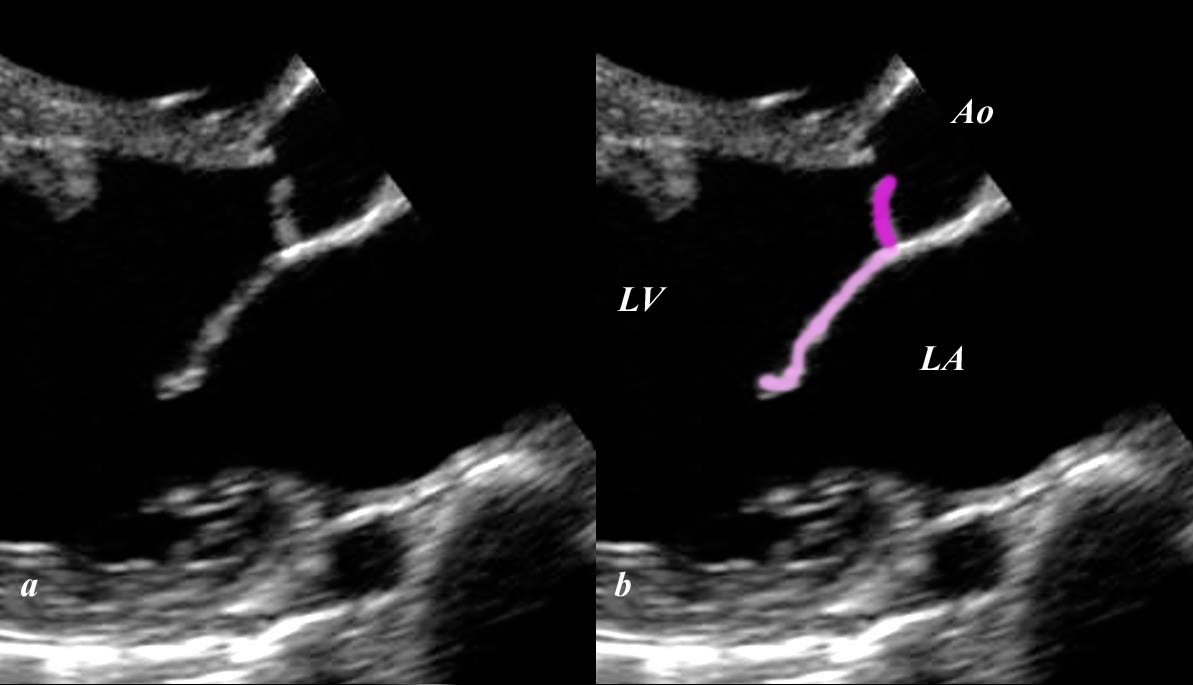
The aortic cusps are anchored to other fibrous portions of the left ventricle. The left coronary cusp and part of the non-coronary cusp are in fibrous continuity with the anterior leaflet of the mitral valve. The right sided portion of the non coronary cusp and the right coronary cusp are intimately related to the membranous portion of the ventricular septum. Thus the ascending aorta is anchored down to the heart via strong fibrous attachments to the mitral valve, membranous septum and to the aortic annulus. In health, the intimate connections enable structural strength and functional integration. On the other hand in disease they provide easy pathways for spread of the disease process.
During the development of the heart the mitral and aortic valve are remote from each other. With resorption of the subaortic infundibulum, the aortic and mitral annulus become connected. This connection is reflected clinically by the fibrous continuity of the anterior leaflet of the mitral valve with the aortic valve.
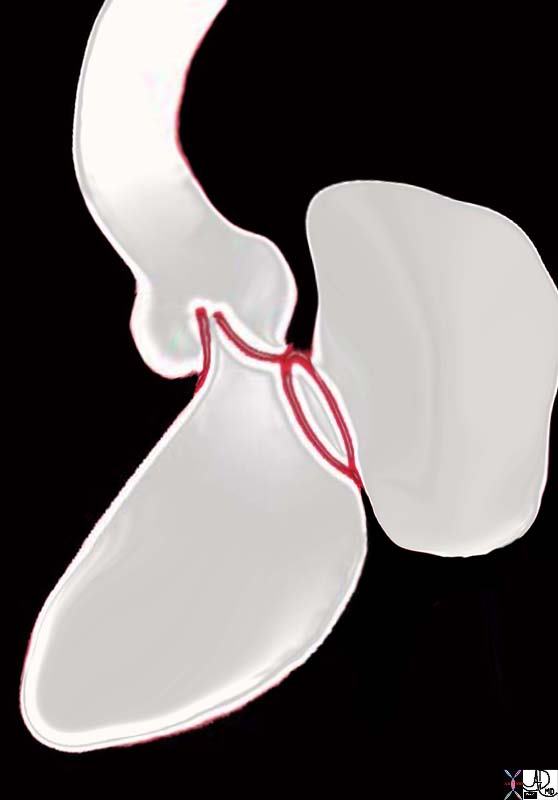 Aortic Valve in Fibrous Continuity with the Mitral Valve Aortic Valve in Fibrous Continuity with the Mitral Valve |
| 06373b05 Courtesy Ashley Davidoff Davidoff drawing |

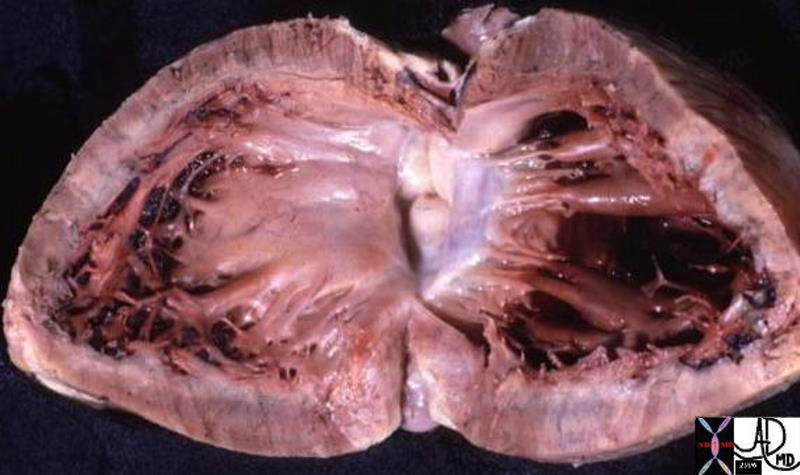
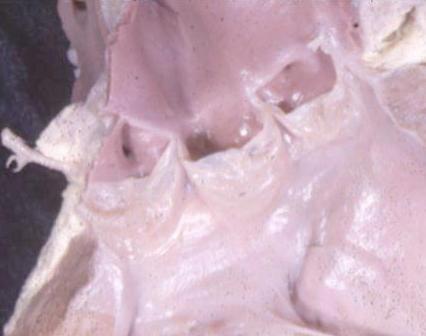
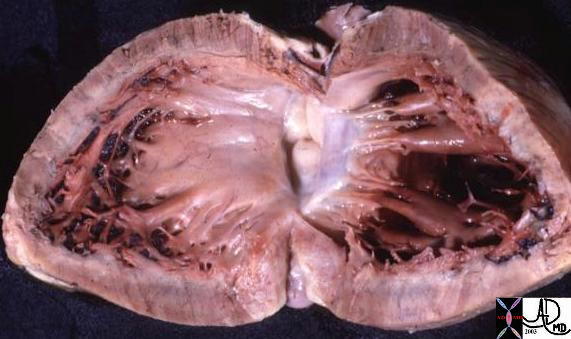 Normal Left Ventricular Outflow Tract (LVOT) Normal Left Ventricular Outflow Tract (LVOT) |
| This anatomic specimen shows the LV with septal side to the left and free wall side to the right. The anterior leaflet of the MV is in full view and note that it is in fibrous continuity with the left and non coronary cusps of the aortic valve.
Courtesy Ashley Davidoff MD 01817 |
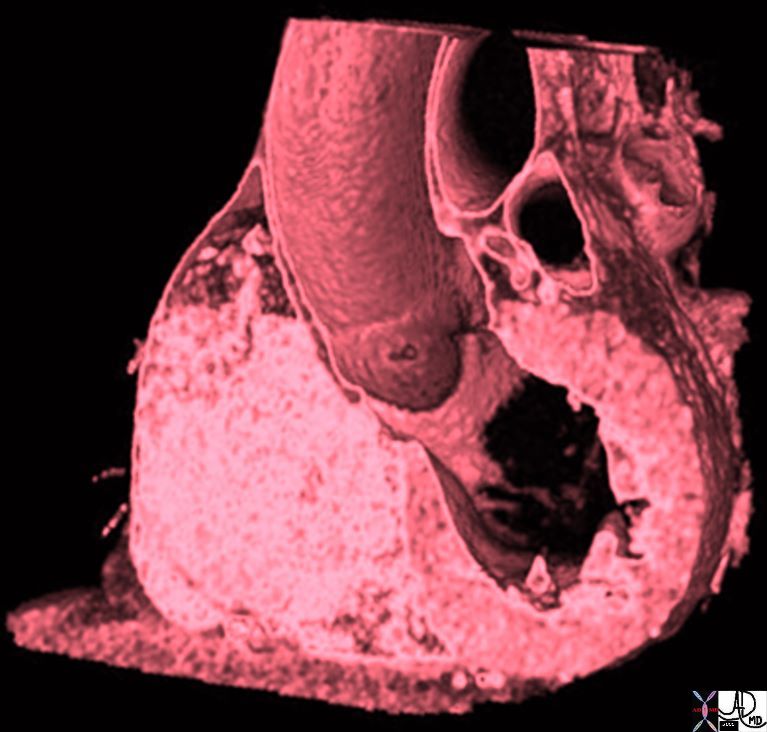
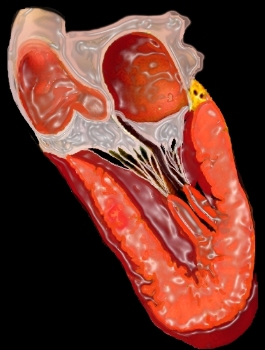
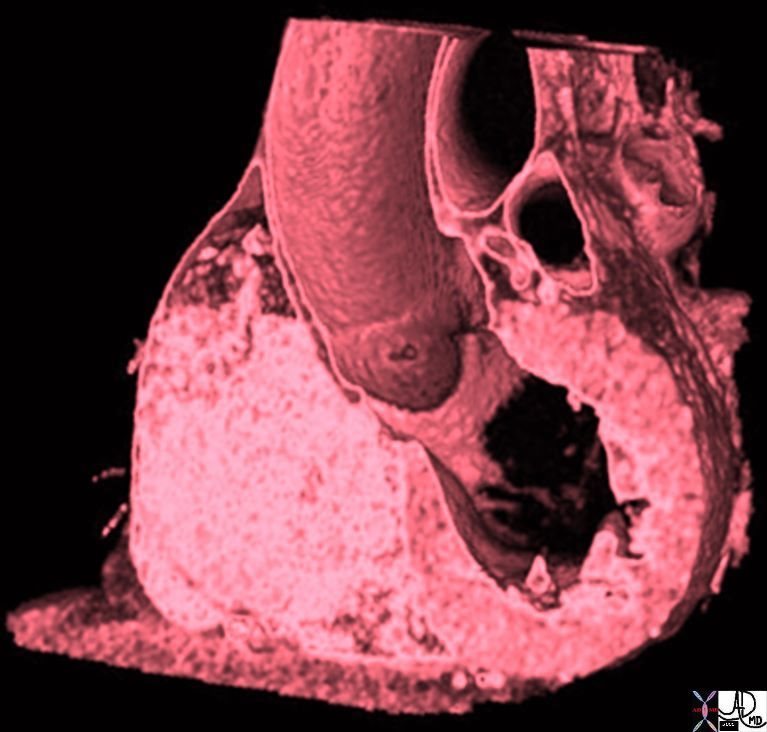
Aortic Valve Connection to the LV and Mitral Valve – Volume Rendered CT |
|
The volume rendered CT shows the anterior leaflet of the mitral valve in continuity with the aortic valve. 47823 CTscan Davidoff MD |
Relationship to the Pulmonary Artery and Pulmonary Valve
The aortic valve lies posterior to, rightward of, and inferior to the pulmonary valve. This relationship is as a result of important embryological changes that occur at the infundibular level. Specifically the subaortic infundibulum resorbs while the subpulmonary infundibulum grows. This action elevates the pulmonary valve and positions it to the left of the aortic valve.
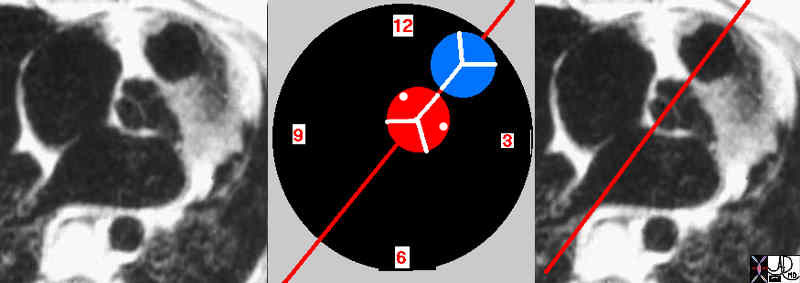
Normal Position and Relationship of the Aorta and Pulmonary Artery |
| 07954eW.800 Davidoff MD |
Functional Considerations
The opening of closing of the aortic valve are predominantly passive , secondary to pressures changes in the cardiac cycle and to the pressure gradient between the LV an aorta .
Opening :During late diastole, as the blood fills the ventricle, a 12% expansion of the aortic root occurs which produces a 20% opening of valve leaflets1. During systole as pressure rises in the ventricular outflow tract, tension across the leaflets lessens and the valve opens rapidly at the beginning of ejection. (More on this can be found in the physiology section of the heart module)
Closing : A principal theory involved in closure is the vortex theory1. The sinus of Valsalva provide a reservoir of blood for small developing vortices. This allows full expansion of the opened valve leaflets, but by maintaining the space between the edge of the leaflet and the aortic wall, it provides for rapid closure at the end of systole by avoiding their sticking to the aortic walls. (More on this can be found in the physiology section of the heart module)
Applied Biology
The close relationship of the aortic valve with the mitral valve is due to the resorbtion of the subaortic infundibulum. When this process becomes deranged, significant consequences result. The absence of this resorbtion results in a group of diseases called the conotruncal abnormalities which include transposition of the great vessels and double outlet right ventricle. The pathogenesis of these diseases will be expanded in the embryology document.
In the acquired categories of disease the proximity of the two valves has relevance in subaortic muscular stenosis where the changes in velocity of blood flow cause suction on the anterior leaflet which results in mitral regurgitation. Ankylosing spondylitis is an inflammatory disease of the sacroiliac joints and spine, and the aortic valve sometimes becomes involved, resulting in thickening of the leaflets and subsequent aortic regurgitation. The close relationship of the anterior leaflet of the mitral valve, the ventricular septum, and the conduction system that runs within the septum, facilitates the spread of the inflammatory process to these structures with potentially significant consequences. In essence, some very important structures are packed close together at this junction, and in health they work well together. However their close proximity in disease has significant multicentric consequence.
Diseases of the aortic valve lead to incompetence (aortic regurgitation) or obstruction (aortic stenosis).
Clinical Aspects
Diagnosis of aortic stenosis is suspected in patients complaining of effort dyspnea, angina or syncope. On examination a weak and slow rising pulse termed pulsus parvus et tardus may be found on carotid palpation. A systolic thrill in the aortic area with radiation along the carotids may be felt. On auscultation an ejection systolic murmur is heard in the aortic area, which at times may have a musical quality and be better appreciated at the apex – Gallavardin phenomena.
Aortic regurgitation presents with symptoms of weakness, dyspnea on exertion , palpitations , orthopnea and angina. Palpation of the radial artery may reveal a pulse characterized by a sharp rise to full expansion followed by immediate collapse – The Corrigan pulse. A positive Hills sign can be demonstrated in most cases – Popliteal cuff systolic pressure exceeding brachial by >20 mm Hg in systole. A mid diastolic murmur of blowing intensity is appreciated in the 2nd aortic space on auscultation with the patient leaning forwards and breath held in expiration.
Imaging
Chest radiography may shown signs of post stenotic dilatation in cases of aortic stenosis along with LV hypertrophy in both stenotis and regurgitation. EKG is non specific with LV hypertrophy and strain pattern. Echocardiography provides functional and anatomical assessment of the valve non invasively and is the diagnostic modality of choice for aortic valve disease. Cardiac catheterization and coronary arteriography may be carried out before aortic valve replacement in select patients.
Aortic Valve Atresia
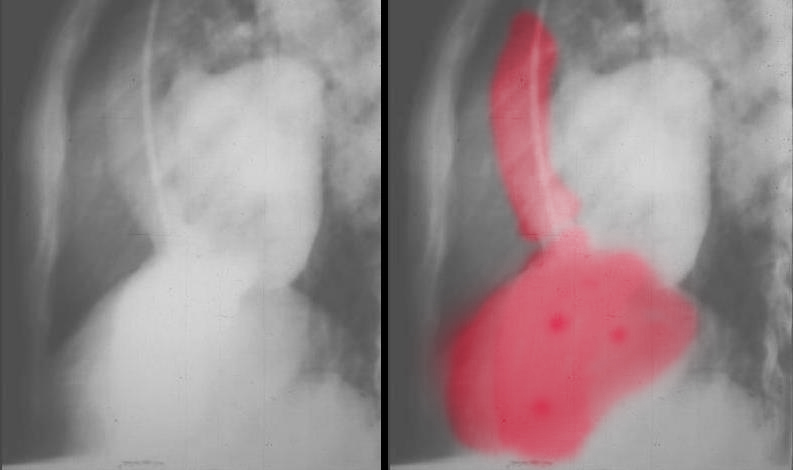
Aortic atresia is the congenital absence of the fetal development of the left ventricle, proximal aorta, and sometimes the mitral valve. This congenital defect results in inadequate systemic oxygen delivery and is usually fatal within 2-3 weeks of life. Diagnostic evaluation using echocardiography is most commonly used. Primary biventricular repair is the most effective surgical treatment of aortic atresia. The Norwood procedure is commonly performed. It involves the reconstruction of the neo-aorta using homograft tissue, pulmonary root tissue and proximal aortic tissue. The procedure requires of cardiopulmonary bypass, aortic cross-clamping and hypothermic circulatory arrest.
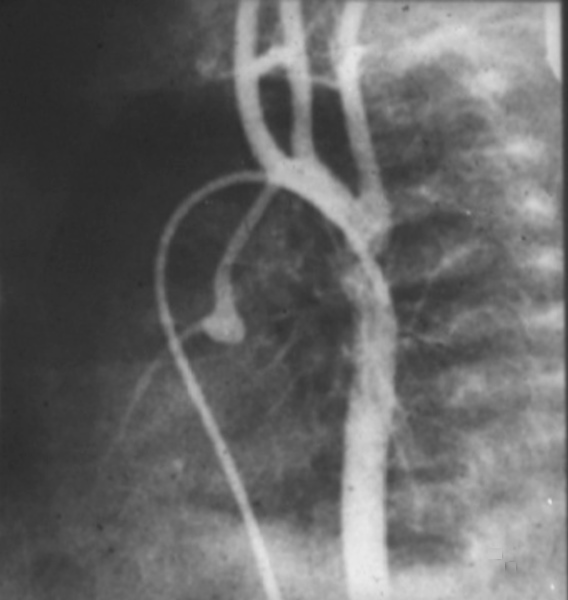
Aortic Valve Atresia in the Setting of Hypoplastic Left Heart Syndrome |
| The aortogram has been performed via a catheter that has entered the right femoral vein, coursed through the right atrium, right ventricle and pulmonary artery entering the descending aorta via a patent ductus arteriosus. There is retrograde filling of the ascending aorta and coronary arteries. The aortic valve is atretic and there is no opening between the LV and ascending aorta. In addition the ascending aorta, arch and isthmus are hypoplastic and there is a post ductal coarctation.
00269b02 Davidoff MD 00269b01 00269b02 00269b03 |
Hypoplastic Left Heart Syndrome (HLHS)
Hypoplastic left heart syndrome is the incomplete formation of the chambers, valves, and or vessels of the left side of the heart. Infants with HLHS are also born with additional defects including ventricular septal defect, patent ductus arteriosus, and narrowing other parts of the aorta. With poor systemic supply of oxygenated blood from the left heart, the ductus arteriosus usually remains open so that blood, albeit deoxygenated blood from the pulmonary circulation, is able to provide some blood to the systemic circulation. Mixing of deoxygenated and oxygenated blood therefore occurs. Infants are born with extremely low oxygen saturation as well as respiratory distress. HLHS can be fatal within hours if untreated. Standard fetal ultrasound is used to diagnose HLHS. This defect is not entirely curable. Various surgical treatments exist in stages including cardiac transplantation if HLHS is severe. The Norwood procedure is commonly performed as the first stage followed by a second procedure which establishes adequate connection for the systemic and pulmonary circulations.
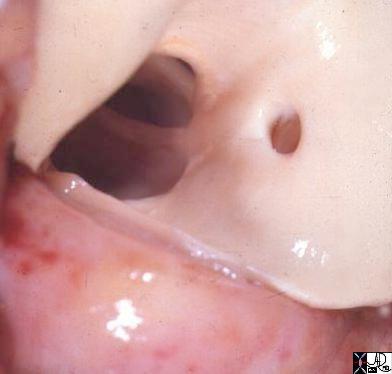
Hypolastic Annulus, Bicuspid Aortic Valve and Malposition of Coronary Ostium |
| 15049 Davidoff MD |
Bicuspid Aortic Valve (BAV)
Bicuspid aortic valve is one of the most common congenital lesions of the cardiovascular system. In this entity only two aortic valve cusps are developed. Patients with BAV commonly have aortic narrowing and regurgitation of variable severity. Most are usually isolated mild lesions but are prone to progressive calcification, and advancing aortic stenosis later in life. With time the gradient increases and the left ventricle hypertrophies to accommodate the increased pressure and work load. The structural distortion of the valve predisposes it to infection and BAV therefore can be complicated by bacterial endocarditis.
Heart murmurs result from either regurgitation or stenosis and are best diagnosed by echocardiography
Surgery with replacement of the valve is indicated when the pressure gradient become high, usually with gradients that are greater than 50-60mmHg across the valve.
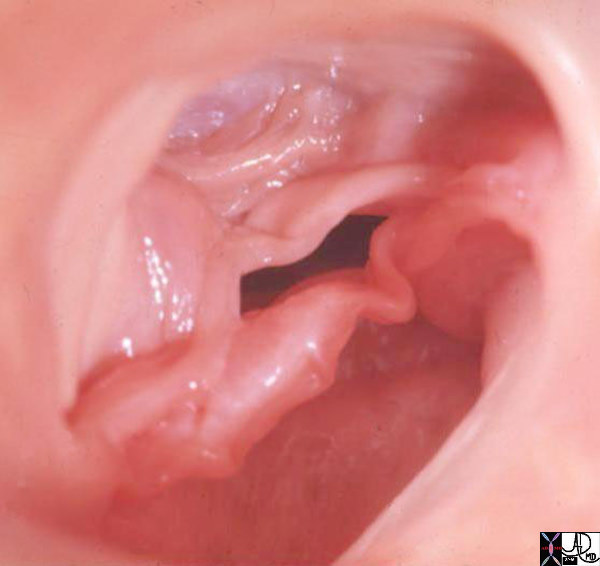 Bicuspid Aortic Valve Caused by Fusion of Two Commissures Between the Right and Non Coronary Cusp Bicuspid Aortic Valve Caused by Fusion of Two Commissures Between the Right and Non Coronary Cusp |
| 07951b Davidoff MD |
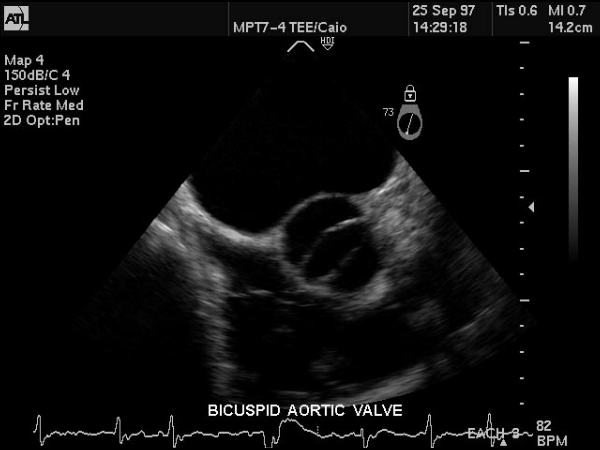 Bicuspid Aortic Valve Bicuspid Aortic Valve |
| This gray scale echo of the heart showing a short-axis aorta left atrial view, and demonstrating the aortic valve with two cusps. The patient has a diagnosis of bicuspid aortic valve which is a congenital condition. Courtesy Philips Medical Systems 33169 code cardiac heart echo aorta bicuspid aortic valve congenital imaging cardiac echo |
|
Normal and Thickened Aortic Valve over Time |
| 07953c02 heart cardiac aorta aortic valve fx normal fx thickened fx bicuspid aortic valve fx calcified fx calcification fusion of the inter-coronary commissures, gross anatomy gross pathology Davidoff MD b Courtesy Henri Cuenoid MD |
Aortic Stenosis – Doming of the Valve |
| The diagram of the left ventricle in LAO (RPO) projection shows a doming of the pliable stenotic valve during systole07969bW.802 Davidoff art |
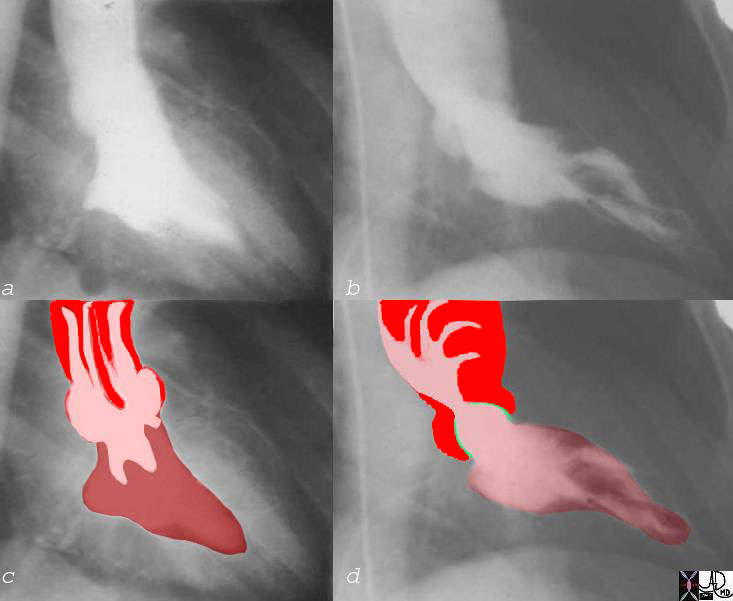
Normal (a,c) and Aortic Stenosis with Post Stenotic Dilatation (b,d) |
| 07969c06 heart aorta left ventricle LV normal anatomy fx hypercontractile ballet slipper ejection fraction = 80% AV aortic valve thickened doming ejection click jet turbulence shear stress post stenotic dilalation dilitation LV angiogram angiography Courtesy Ashley Davidoff MD aortic valve 07969c03 07969c02 07969c04 |
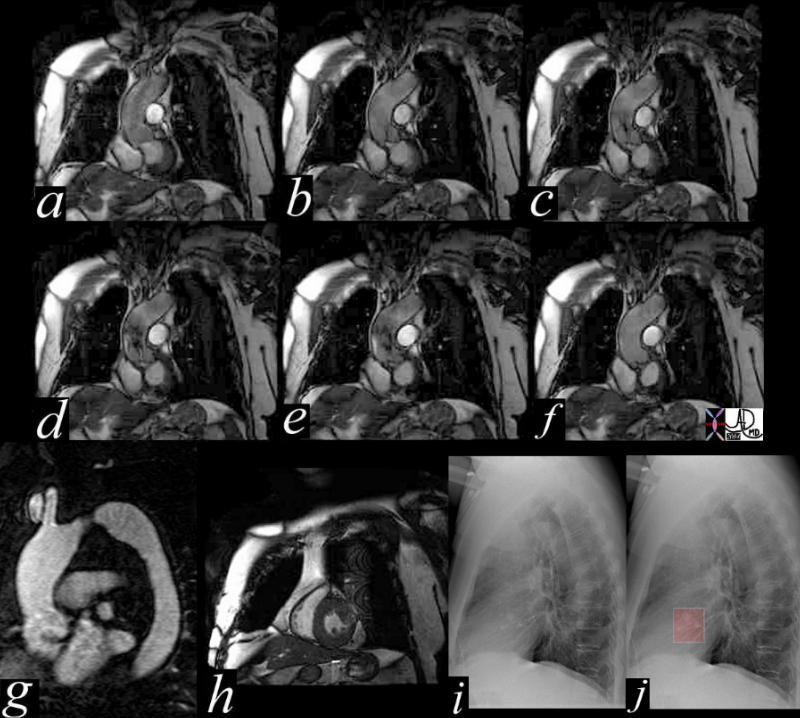
Aortic Stenosis – Turbulence Shown by MRI |
| This series of coronal MRI images of the aortic valve (a-f) show phases from diastole (a) through systole (b,c,d,e) with a narrow (b,c) and then turbulent jet, (d,e) back to diastole (f) Image g shows a thickened valve, while the short axis of the LV (h) shows LV hypertrophy. The plain film of h and i highlight the calcific nature of the valve. The diagnosis is aortic valve stenosis. Courtesy Scott TSai MD 38871c01 code cardiac heart aortic valve AS LVH calcification calcified imaging radiology MRI CXR plain film |
Aortic Sclerosis
Aortic sclerosis is a degenerative disease commonly seen in older patients. It results in the progressive calcification and narrowing of the aortic valve. These effects cause heightened pressures in the left ventricle. The detection of a evolving ejection murmurs, left ventricular hypertrophy, and aortic valve calcification in an elderly patient suggests the diagnosis, and the diagnosis can be confirmed with echocardiography and CTscan. If severe, aortic valve replacement is required.
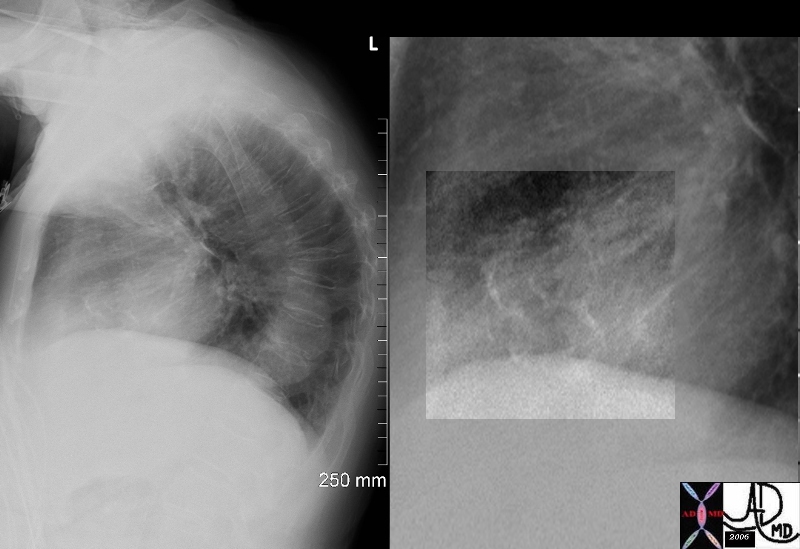
The calcification of the aortic valve and or annulus is highlighted in the second image. The osteopenia, compression fractures, and kyphosis are frequently associated findings, particularly in elderly women.
Ashley Davidoff MD TheCommonVein.net 47368c02
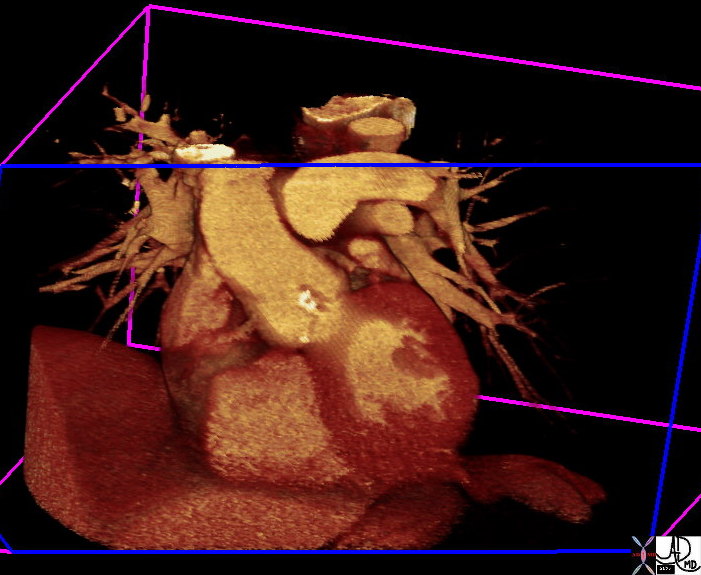
Calcific Aortic Stenosis and Sclerosis with LVH |
|
The coronal reformat shows a thick walled left ventricle and calcification of the aortic valve. (white irregularity on the valve) This finding is characteristic of aortic stenosis. 39699.800 Courtsey Ashley Davidoff MD |
Aortic Regurgitation
Aortic Regurgitation is a malfunction of the aortic valve resulting in the improper closure of the leaflets. This results in blood leakage or backflow. Aortic regurgitation is caused by congenital abnormalities such as bicuspid aortic valve or acquired abnormalities such as rheumatic heart disease or bacterial endocarditis. Then hemodynamic consequence of aortic regurgitation is volume overload of the left ventricle. Mild cases are treated with medically. If severe, aortic valve replacement surgery is necessary.
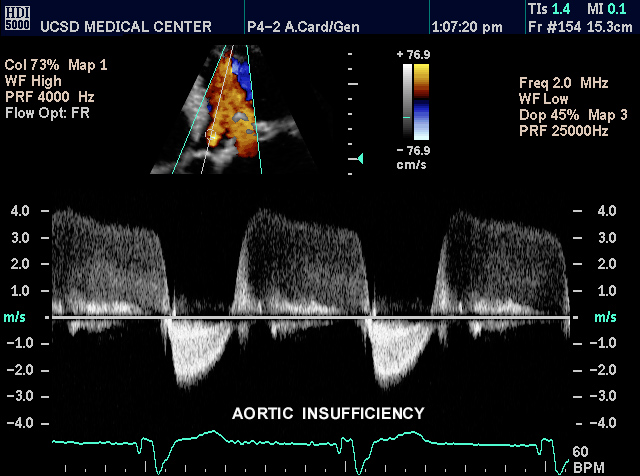 Aortic Insufficiency |
| This color flow doppler echo of the heart with pulse flow interrogation at the level of the aortic valve showing a color jet of regurgitation of the valve. The patient has a diagnosis of aortic regurgitation Courtesy Philips Medical Systems 33118 code cardiac heart AO valve AI echo pulse doppler color imaging cardiac echo |
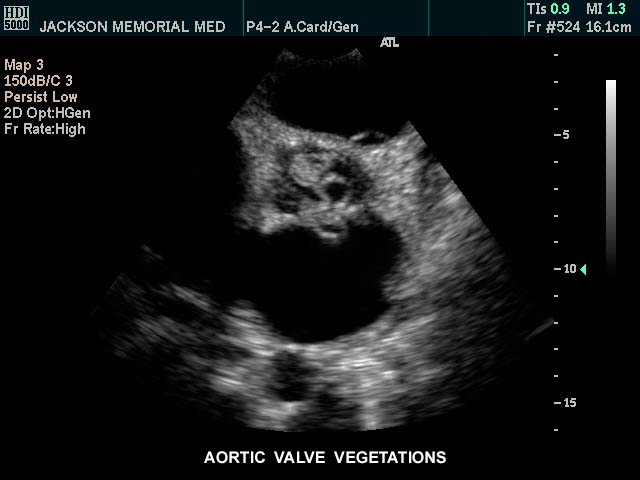
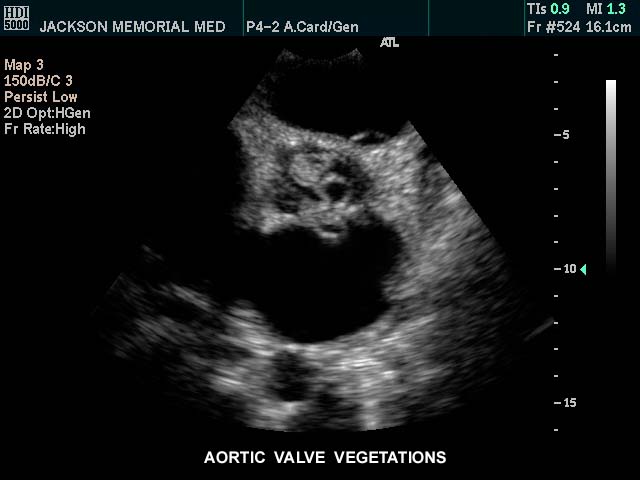 This gray scale echo of the heart showing a short-axis aorta left atrial view,, and demonstrating vegetations on the aortic valve. The patient has a diagnosis of bacterial endocarditis. Courtesy Philips Medical Systems 33126 code cardiac heart echo AO valve vegetations SBE bacterial endocarditis infection imaging cardiac echo Aortic Valve Vegetations |
| This gray scale echo of the heart showing a short-axis aorta left atrial view,, and demonstrating vegetations on the aortic valve. The patient has a diagnosis of bacterial endocarditis. Courtesy Philips Medical Systems 33126 code cardiac heart echo AO valve vegetations SBE bacterial endocarditis infection imaging cardiac echo |